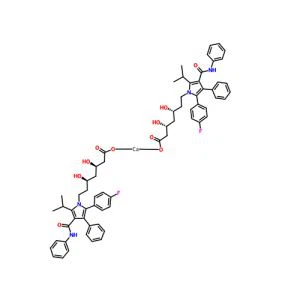
Atorvastatin calcium Basic information
|
Product Name: |
Atorvastatin calcium |
|
Synonyms: |
Atorvastatin Calcium Anhydrous;Calcium of atorvastatin;Atorvastatin ;Atorvastatin aMorphous;atorvastatine calciuM salt;(βR,δR)-2-(4-Fluorophenyl)-β,δ-dihydroxy-5-(1-Methylethyl)-3-phenyl-4-[(phenylaMino)carbonyl]-1H-pyrrole-1-heptanoic Acid CalciuM Salt;Atorvastatin HeMicalciuM;Sortis |
|
CAS: |
134523-03-8 |
|
MF: |
C66H68CaF2N4O10 |
|
MW: |
1155.36 |
|
EINECS: |
200-659-6 |
Atorvastatin calcium Chemical Properties
|
Melting point |
176-178°C |
|
storage temp. |
Store at +4°C |
|
solubility |
DMSO: ≥10mg/mL |
|
form |
white powder |
Atorvastatin calcium Usage And Synthesis
|
Mode of action |
Atorvastatin takes effect through selectively and competitively inhibiting the hepatic enzyme HMG-CoA reductase, which is responsible for converting HMG-CoA to mevalonate in the cholesterol biosynthesis pathway. This results in a subsequent decrease in hepatic cholesterol levels. Decreased hepatic cholesterol levels stimulates upregulation of hepatic LDL-C receptors which increases hepatic uptake of LDL-C and reduces serum LDL-C concentrations. |
|
Chemical Properties |
White Crystalline Powder |
|
Uses |
The primary uses of atorvastatin is for the treatment of dyslipidemia and the prevention of cardiovascular disease. |
Atorvastatin calcium Specification
|
Character |
A white or almost white?crystalline powder |
||
|
Identification |
A: IR. |
||
|
water |
3.5~5.5 |
||
|
Ca % |
3.0~3.6 |
||
|
Particle size |
D90≤10 µm |
||
|
Heavy Metals(ppm) |
≤20 |
||
|
Impurity |
Impurity E |
≤0.3% |
|
|
Impurity A |
≤0.3% |
||
|
Impurity?B |
≤0.3% |
||
|
Impurity C |
≤0.3% |
||
|
Impurity D |
≤0.1% |
||
|
Other individual unknown Impurity % |
≤0.1% |
||
|
Total impurity |
≤1.0% |
||
|
Assay % |
98.0-102.0 |
||
http://www.jecibiochem.com/